UPDATE ON LIVER DAMAGE FROM HERBALIFE, BLACK COHOSH and KAVA
neil.burman@gmail.com
16 March 2012
it is comforting to note that there have been no new reports the past year of toxicity from these products.
In fact some lab work seems to favour black cohosh for cancer prevention;
Anticancer Res. 2012 Jan;32(1):21-30. Chemopreventive potential of black cohosh on breast cancer in Sprague-Dawley rats. Einbond LS, Soffritti M, Degli Esposti D, Tibaldi E, Lauriola M, Bua L, He K, Genovese G, Su T, Huggins L, Wang X, Roller M, Wu HA. Columbia University, HHSC-1518, 701 W. 168th Street, New York, NY 10032, USA. lseinbond@gmail.com
This study examines the chemopreventive potential and action of the
herb black cohosh on Sprague-Dawley rats. CONCLUSION:Our results suggest
that black cohosh may have chemopreventive potential for mammary
cancer.
while Herbalife has published objective rebuttals of toxicity from
Herbalife other than in isolated countries where irregular ingredients
were used in local manufacture:
World J Hepatol. 2011 Oct 27;3(10):275-7. Revisiting acute liver injury associated with herbalife products. Appelhans K, Smith C, Bejar E, Henig YS
Herbalife International of America Inc., Torrance, CA 90502, United
States. In the November 27, 2010 issue of the World Journal of
Hepatology (WJH), three case reports were published which involved
patients who had consumed various dietary supplements and conventional
foods generally marketed as weight loss products. The reference to
Herbalife products as contaminated and generally comparable to all
dietary supplements or weight loss products is not scientifically
supported. The authors provided an insufficient amount of information
regarding patient histories, concomitant medications and other
compounds, dechallenge results, and product specifications and usage.
This information is necessary to fully assess the association of
Herbalife products in the WJH case reports. Therefore, the article does
not objectively support a causal relationship between the reported cases
of liver injury and Herbalife products or ingredients.
Pharmacoepidemiol Drug Saf.
2012 Mar;21(3):333-4. doi: 10.1002/pds.3203. Misconceptions regarding
the association between Herbalife products and liver-related case
reports in Spain.
Appelhans K, Frankos V, Shao A. Source Product Compliance and Safety, Herbalife International of America, Inc, Torrance, CA, USA.
Since June 2010 there have been no new cases of toxicity from Black Cohosh, herbalife or kava reported on Pubmed.
BLACK COHOSH: BEGGING THE QUESTION OF INDICATION- NEED.
The new literature analysis Suspected black cohosh hepatotoxicity: no evidence by meta-analysis of randomized controlled clinical trials for isopropanolic black cohosh extract by Naser et al from Yale , and Germany(the main producer of black cohosh BC products)- begs the question.
As this colunm has previously reviewd about BC, women have died from or needed liver transplants after taking it.. Hence most Authorities have Black Box warning requirements Recurrences of liver reaction have been reported on rechallenge. These scattered cases can be argued away on metanalysis, but they cannot be ignored. One death or acute liver failure is unacceptable when BC is never an essential drug without other safe options.
Another study also published now (Wang ea from the FDA Centre for Drug Evaluation ) http://www.ncbi.nlm.nih.gov/pubmed/20920542 contradicts the German metanalysis with more basic toxicological data: “Computational analysis of positively predicted constituents showed … specifically, protocatechuic acid from black cohosh… predicted positive for liver toxicity endpoints also confirmed with literature findings”
Black cohosh is not physiological hormone replacement, BC is recommended by its proponents solely for menopause symptoms (it has no other benefits) for up to 6 months.
So why risk, use black cohosh at all?
Appropriate balanced hormone replacement – preferably human hormones, not xenohormones ie hormones not found in the healthy women, and not by swallowing it- is indicated permanently in all women .
As previouslly pointed out in this column, the International Menopause Society has summed it up in putting approriate HRT as the main agent(s) for menopause symptoms as well as for its permanent multisuystem benefits; and the human hormone gamma-aminobutyric acid GABA as the only alternative that is both safe and cleearly proven better than placebo for improving both hot flashes and sleep, anxiety. Used appropriately and with sensible monitoring and dose titration, all such hormone balance has no longterm risks.
MDICOLEGAL LIABILITY: under the new Conumae Protection Act CPA in South Africa, the pendulum has gone ridiculously too far. irrespective of the onus on manufacturers and promoters of any product, the onus is on the end-prescriber, end-dispenser to warn consumers of potential risks, and any consumer claim for consequent damages is legally against only the final and retail supplier.
So no supplier of black cohosh is protected against consequent liability unless he gets a signed waiver from the purchaser after the recorded warning about its potential toxicity.
16 June 2010
there are no new adverse toxicity reports on Hebalife, black cohosh or kava so far in 2010 .
Both Herbalife, and black cohosh products, remain marketed and in demand in South SAfrica.
There are no new serious adverse reports or concerns published on Pubmed or Google about Herbalife products in 2009.
On Pubmed there were 2 new cases of liver and coagulation problems associated with black cohosh in mid2009, from a Germanic and an Italian institute; and 14 hepatitis cases associated with kava ingestion confirmed from around the world .
4 Jan 2009
This review is not about benefit of black cohosh (independent trials show none for menopause symptoms) or Herbalife (trials support that Herbalife is indeed a weight loss aid), or kava (it is a confirmed anxiolytic analgesic euphoriant); but about toxicity potential however rare – considering that none of these products can make any claim to being a necessity.
Contamination aside, there are no new relevant reports on Herbalife the past two months on Pubmed,
but indeed 4 new reports on black cohosh; and one on kava.
Lessons for black cohosh and Herbalife may be learnt from kava. Kava-kava was hastily banned eg in Europe and South Africa early this decade owing to reputed association with hepatotoxic deaths. But on careful study these toxicity claims appear to be uncertain.
The claimed benefits of kava are analgesic, euphoriant and relaxant, without addiction potential. The four trials of Kava (between 1991 and 2003, in Italy and Germany) confirmed that kava has anxiolytic benefits in the menopause syndrome.) . The hepatotoxicity (not reported from the source – Polynesia - unless taken by alcoholics) was reported largely from western countries, where commercially sold kava extract was apparently differently extracted, and from the aerial leftovers of the kava; whereas in Polynesia it is extracted only from the root. A recent website from the NIH shows it is not banned there, but expresses much caution.
A careful analysis of kava hepatotoxicity by Teschke ea in Germany last month again finds little evidence of toxicity if kava is taken from a reputable manufacturer at prescribed dose and for short duration – as applies equally to alcohol, and most drugs.
Herbalife
An objective NICUS Nutritional Institute of University of Stellenbosch Report critical of Herbalife is quoted verbatim in the Summer 2008 Newsletter of the Association of Dieticians of SA. NICUS- a world authority in Nutrition – confirms it stands by this report. It could thus be taken as a directive (to condemn Herbalife)- to dieticians for whose professional advice some patients wrongly substitute diet supplements; where these modalities- careful professional diet advice and counselling, and supplements- are actually complementary..
But there does not appear to be any more evidence to condemn Herbalife than there was a year ago. As far as Pubmed and Google reveals, the reports [to date end of 2008 on Pubmed) of adverse effects were from 4 discrete European regions [ Iceland; Switzerland; Spain & Israel) , apparently wth locally formulated Herbalife, not the USA main factory product, leading to assumption of a local production fault. There appear to have been 2 cases of liver failure in some 33 affected patients, in one of whom liver transplant became necessary but the patient died.. .
One cannot condemn all babyfood because some is deliberately adulterated with melamine by ruthless Chinese sham factories. Commercial babyfood is arguably a necessity for many.
We await an updated rebuttal from Herbalife in the new year- but there does not seem to be anything for them to add to their rebuttal of last year..There has been no published evidence to justify update on Herbalife during 2008.
The accusation (by one patient, and a convicted fraudster, Minkow) of a claimed lead-contaminated Herbalife batch in California has, strangely, generated no updates for months now- but on the wiki herbalife update , it says "In August 2008, Minkow retracted all accusations against Herbalife and removed any mention of the company from his Web site.[30]” – there is no report of whether Herbalife bought his silence or not , which is a pity- see also. ; but see also heavy flak against Minkow , suggesting that his whole campaign was a successful bear scam to profit from Herbalife shares.
The current Wiki Herbalife review also quotes a new trial validating benefit for weightloss. Caveat Emptor.
It may be asked why a Nutritional authority like NICUS: warns against Herbalife but not against the potentially fatal black cohosh. New independent analyses are both for and against them:
ie
Black Cohosh:
Analyses of case reports from Univ Florida (Palacio 2009) and Italy (Borrelli 2008) recommend caution about black cohosh for humans in view of adverse case reports; while a German analysis (Teschke 2008) exonerates black cohosh in every single case till then.
A trial from USA (Davis 2008) found that “black cohosh significantly increased the incidence of lung metastases in tumor-bearing mice compared with mice fed the isoflavone-free control diet”.
Clearly the valid divergence of opinion comes down to complex statistics of probability.
Black cohosh has been associated with severe liver failure and transplantation in a number of women on a number of continents, for which reason the local Health Products Association, like all responsible authorities , finally agreed and issued recommendations that Black Cohosh label must be black-boxed- there is no justification for it’s sale as a useful product, unlike the role than can be argued for food substitute powders.
So why should anyone use black cohosh for menopause symptoms (when it’s benefit seems to be largely placebo, with grave doubt about safety) , when there are proven safe symptom relievers eg GABA (no adverse reports); or lowdose balanced appropriate parenteral human sex hormones (no adverse reports, and have numerous longterm multisystem benefits- which neither black cohosh nor GABA can claim..).
This review is not about benefit, but safety. Regulators remain silent about drastically curtailing sale of the most lethal substances in widespread unregulated (and unnecessary) use- paracetamol, other nonsteroidal anti-inflammatories, statins for uncomplicated mild-to-moderate lipidemia, alcohol, tobacco and sugar, and pollution of everything by industrial adulteration with synthetic (often estrogenic) endocrine disruptors and virtually all fast foods with cornstarch and/or sugar.
The hysterical approval of diethylstilbestrol DES by the FDA by 1950, and for a massive 1950 maternity trial against all evidence (even though it’s toxicity was recognized by 1953, it’s sale anywhaere was finally banned only 20 years later) continues to torment the original myriads of guineapig women, their children and now grandchildren. These scandals are dictated by individual opportunist, corporate and governmental greed, and indifference to medical evidence and prevention.
The analogy for black cohosh, kava and Herbalife is perhaps:
#the ~20year delays before the FDA would licence the lifesaving lithium salt, and metformin, in USA;
#the ~5year hysteria over HRT after the Women’s Health Initiative – when the over-estimated risks of inapproriate use of OHT in elderly women were stupidly and harmfully (for thousands of women) extrapolated to young women and other HRT preparations;
and
#the melamine- baby milk formula catastrophe – the problem for the latter was exclusively some contaminated babyfood batches made in China especially for the lucrative export market. .
The jury can thus be considered as still out on both black cohosh, kava and Herbalife, until manufacturers of commercial products (not traditional preparations of eg kava and black cohosh taken by residents who grow these) can produce evidence, confirm that the risk was limited to specific batches of the commercial product and adherence to accepted recommendations, and not due to other possible risk factors.
( Black Cohosh: Side effects in wikipedia )
Studies on human subjects who were administered two commercially available black cohosh preparations did not detect estrogenic effects on the breast.[15]No studies exist on long-term safety of black cohosh use in humans.[20] In a transgenic mouse model of cancer, black cohosh did not increase incidence of primary breast cancer, but increased metastasis of pre-existing breast cancer to the lungs.[21]
Liver damage has been reported in a few individuals using black cohosh,[2] but many women have taken the herb without reporting adverse health effects,[22] and a meta-analysis of several well-controlled clinical trials found no evidence that black cohosh preparations have any adverse effect on liver function.[23] Despite a lack of conclusive evidence for a link between black cohosh and liver damage, Australia has added a warning to the label of all black cohosh-containing products, stating that it may cause harm to the liver in some individuals and should not be used without medical supervision.[24] Other studies conclude that liver damage from use of black cohosh is unlikely,[25] and that the main concern over its safe use is lack of proper authentication of plant materials and adulteration of commercial preparations with other plant species.[26]
Reported direct side-effects also include dizziness, headaches, and seizures; diarrhea; nausea and vomiting; sweating; constipation; low blood pressure and slow heartbeats; and weight problems.[27]
Because the vast majority of black cohosh materials are harvested from plants growing in the wild,[2] a recurring concern regarding the safety of black cohosh-containing dietary supplements is mis-identification of plants causing unintentional mixing-in (adulteration) of potentially harmful materials from other plant sources.[2]
================================
Herbal hepatotoxicity: a tabular compilation of reported cases.
Source
Department of Internal Medicine II, Division of Gastroenterology and Hepatology, Klinikum Hanau, Academic Teaching Hospital of the Medical Faculty of the Goethe University, Frankfurt/Main, Germany.Abstract
BACKGROUND:
Herbal hepatotoxicity is a field that has rapidly grown over the last few years along with increased use of herbal products worldwide.AIMS:
To summarize the various facets of this disease, we undertook a literature search for herbs, herbal drugs and herbal supplements with reported cases of herbal hepatotoxicity.METHODS:
A selective literature search was performed to identify published case reports, spontaneous case reports, case series and review articles regarding herbal hepatotoxicity.RESULTS:
A total of 185 publications were identified and the results compiled. They show 60 different herbs, herbal drugs and herbal supplements with reported potential hepatotoxicity, additional information including synonyms of individual herbs, botanical names and cross references are provided. If known, details are presented for specific ingredients and chemicals in herbal products, and for references with authors that can be matched to each herbal product and to its effect on the liver. Based on stringent causality assessment methods and/or positive re-exposure tests, causality was highly probable or probable for Ayurvedic herbs, Chaparral, Chinese herbal mixture, Germander, Greater Celandine, green tea, few Herbalife products, Jin Bu Huan, Kava, Ma Huang, Mistletoe, Senna, Syo Saiko To and Venencapsan(®) . In many other publications, however, causality was not properly evaluated by a liver-specific and for hepatotoxicity-validated causality assessment method such as the scale of CIOMS (Council for International Organizations of Medical Sciences).CONCLUSIONS:
This compilation presents details of herbal hepatotoxicity, assisting thereby clinical assessment of involved physicians in the future==============================
A rare cause of drug-induced hepatitis in an immunocompromised patient and the role of glutathione.
Source
Viplove Senadhi, Division of Gastroenterology and Hepatology and Brater Scholar, Indiana Institute for Personalized Medicine, Indiana University School of Medicine, Indianapolis, IN 46202, United States.Abstract
The Food and Drug Administration (FDA) has issued a warning on numerous herbal drugs, including many popular products at General Nutrition Centers (GNC), regarding unstudied hepatotoxicity. There have been recent reports of GNC products such as hydroxycut and herbalife, causing drug-induced hepatitis. Herbal medications are over-the-counter products and are not investigated thoroughly by the FDA. Given that the most common outpatient laboratory abnormality is elevated liver transaminases, a sign of hepatocellular toxicity; it is not surprising that some of these products end up causing hepatic dysfunction, especially when taken in large volume. There are numerous herbal supplements that are hepatotoxic, however, these medications have a much more significant effect in human immunodeficiency virus (HIV)/ acquired immune deficiency syndrome patients, which is secondary to depleted glutathione. We present a rare case of drug induced hepatitis secondary to herbal medications used to treat HIV and elucidate the role of glutathione depletion in immunocompromised patients.================================
Continuous reporting of new cases in Spain supports the relationship between Herbalife® products and liver injury.
Pharmacoepidemiol Drug Saf. 2011 Oct;20 (10):1080-7. doi:10.1002/pds.2180. Epub 2011 Jul 12.
Manso G, López-Rivas L, Salgueiro ME, Duque JM, Jimeno FJ, Andrade RJ, Lucena MI.
Source
Centro de Farmacovigilancia de Asturias, Facultad de Medicina, Universidad de Oviedo, Oviedo, Spain. gmanso@uniovi.es
Abstract
PURPOSE:
Previous publications have linked Herbalife® products to hepatotoxicity. The identification of earlier cases in which the culprit agent could not be established raised the hypothesis of a possible contamination of some specific batches of Herbalife products.METHODS:
We searched the Spanish Pharmacovigilance Centres' database of adverse reactions for reports of liver injury associated with the use of Herbalife products from 2003, when the first case was submitted, through September 2010.RESULTS:
The search resulted in 20 reports of liver damage (mean age, 49 years; 16 women), with 12 patients (60%) requiring hospitalization. Hepatocellular damage predominated, and nine (53%) of the hepatocellular cases with bilirubin values were jaundiced, fulfilling the Hy's law criteria, which increases the risk for serious outcomes. Two patients experienced a positive rechallenge. One patient developed cirrhosis, whereas all the others recovered. Causality assessment by the Karch and Lasagna modified algorithm showed a category of definite in 1 case, probable in 14, and possible in 5. Analysis of the different Herbalife products that each patient had taken did not enable us to identify any commonly known hepatotoxic ingredient.CONCLUSIONS:
Our results support the relationship between the consumption of Herbalife products and hepatotoxicity, underscore the concern regarding the liver-related safety of this dietary supplement, and emphasize the need to establish further regulatory measures.
BACTERIA IN HERBALIFE DAMAGES LIVER
Two years ago liver doctors sounded the alarm about cases of liver damage
in users of Herbalife products. They were unable to say which component
in Herbalife products was causing the liver damage, but researchers at
the University of Bern made an attempt to do so in an article published
this year in the Journal of Hepatology. They found the bacteria Bacillus subtilis in Herbalife products.

|
In the article the doctors discuss two cases of people who became ill
after using Herbalife products: a man aged 78 and a woman aged 50. The
man's urine
had turned dark brown, he had hepatitis and had been feeling unwell for
a couple of weeks. He had been using the Herbalife F1 Shake [Strawberry
and Cappuccino flavours] for three years on his daughter’s advice [she
was a Herbalife salesperson], as well as various other medicines.
According to the analyses, the man had a liver complaint. When the
doctors took him off the shake his blood values recovered, but they
deteriorated again later. The doctors gave the man corticosteroids and ursodeoxycholic acid, after which he recovered completely. You can see the fluctuations in the man’s blood values in the graph below.

The woman sold Herbalife supplements. She took half a dozen different
Herbalife supplements, including the Personalized Protein Powder Mix
Formula 3. She had stomach pain
and hepatitis and the doctors found signs of liver damage in her blood.
They got the woman to stop taking the supplements and the graph below
shows how the liver values in her blood improved.
The researchers examined samples from the livers of the man and the
woman and found signs of damage in both. The doctors turned the
supplements that the men and women had used inside out, but found no
contaminants: no heavy metals, no pesticides, no antibiotics, nothing.
But when they examined the Herbalife products for micro-organisms, they
did come across something. The meal substitutes the man and woman had
been using contained the bacteria

|
Hmm.
Honestly speaking, we’re not sure what to make of this. As far as we know, B. Subtilis is found in pretty much everything, and it’s not particularly dangerous. [Wikipedia]
Some studies even regard it as a probiotic. It would seem pretty unlikely then that B. subtilis is the cause of such serious liver damage.
Source: J Hepatol. 2009 Jan;50(1):111-7.
Bodybuilder drugs his liver to oblivion – twice
A medical case study that will give you goose bumps, this report from
doctors at the US Massachusetts General Hospital. The article tells
the story of a 27-year-old competitive bodybuilder and part-time
policeman who turned up at the Emergency Department.
The man’s been using anabolics for five years, but now he's got stomach
pains and is nauseous. The doctors do blood tests and discover
that the man’s liver is no longer functioning properly. For a start, the
concentration of the enzyme ALT is 2457 units per litre: 35
is the healthy maximum. The bodybuilder’s blood is syrupy and his
haematocrit level is fifty percent.
The bodybuilder says he injects nandrolone and takes androstenedione.
The scan the doctors make of his liver shows a real mess, shown in
Picture A below. The light patches are swellings, the biggest of which
measures 10 x 10 cm.

The doctors operate and discover that the liver is in an even worse
state than they had feared. The organ contains swellings that are filled
with blood. Peliosis hepatis is the doctors’ diagnosis, the same
complaint as killed Andreas Muenzer.
The doctors also found a swelling with a diameter of 23 cm. This kind of
swelling doesn’t usually get bigger than 3 cm. The doctors decide
to amputate part of the liver.
After the operation the bodybuilder recovers and is allowed home. When
the doctors make another scan three months later they see that the
swellings
in the remaining part of the liver have shrunk by 40 percent, Picture B
above. The bodybuilder thinks it's time to start doping again, and says
he’s
going to start again with androstenedione.
After three years the bodybuilder returns to the doctors. Six weeks
previously he had treated himself to injections of a nandrolone
derivative.
And bingo, he’s got stomach pains again.
Three days after admission to the hospital, the bodybuilder’s heart rate
starts to increase. When the doctors make another scan – the picture
above on the right – they see that his liver is in a bad way again. The
swellings have started to grow again, and the liver has a protuberance
that has started to bleed.
The doctors discuss briefly whether they should give the guy a new
liver, but reject the idea. It would be a waste: he would only destroy
it again
with pills and injections. Alcoholics are not given a new liver either.
So they just stop the bleeding and send the bodybuilder home again.
Thinking about it, we find it difficult to believe that the bodybuilder
was only using deca and androstenedione. True, a number of cases have
been
published of bodybuilders who, as the result of a mild course of deca,
developed a blood clot the size of a brick in their stomach, or became
psychotic after one single deca injection. But to be honest, we don’t
believe those studies.
Moreover, neither androstenedione nor nandrolone is harmful to the
liver. Laboratory research on liver cells has shown this. And, by the
time the
doctors were seeing their patient, George Bush had already outlawed
androstenedione. Although androstenedione is still on the market,
bodybuilders
hardly ever use it. Why would they? In countries like the US there are
much more effective legal anabolic steroids available. But among these,
there are a number of substances that are notoriously bad for the liver.
The most dangerous is Superdrol, an anabolic that was developed at the
end of the fifties by Syntex. Doctors have recorded damaged livers and
destroyed kidneys in users of Superdrol more than once.
This is what makes us suspect that the bodybuilder in this case study
used stronger stuff than androstenedione and deca. Bodybuilders are
often
not entirely honest about what they have been using when they visit a
doctor about medical problems. Even when being honest could have saved
their
life.
Sources:
World J Gastroenterol. 2008 Jul 28;14(28):4573-5.
Bodybuilder gets jaundice from creatine and protein supplements
A healthy man of 27, a fanatical bodybuilder, developed liver damage and jaundice after using sports supplements
containing creatine and whey protein. Liver specialists at The Mount Sinai Medical Center in New York describe the
case in Seminars in Liver Disease.
The man in the case study had no pain and did not feel unwell, but because he showed signs of jaundice he went to
hospital. There the doctors discovered that his blood contained high levels of bilirubin, a substance that should
have been removed by the liver. The man’s blood also contained high amounts of the enzyme alkaline phosphatase and
creatinine. The first substance can be an indication of liver malfunction. The second is a waste product of
creatine.
In people with jaundice, the skin and whites of the eyes turn yellow [see pic below], but this is not necessarily
cause for alarm. Bilirubin is not a dangerous substance, and may even be an endogenous antioxidant. But jaundice is a
sign that something is wrong with the liver, and it therefore needs to be taken seriously.
When the doctors examined the bodybuilder’s liver they saw that the organ was no longer removing bile. The ducts that
should have been doing so were blocked. The scientific term for this is cholestasis.
The bodybuilder had been taking creatine for nine months and whey protein for one month. The doctors thought that this
might be responsible for his condition. When the man stopped taking the supplements, his liver returned to normal and
the jaundice disappeared. If doctors come across healthy athletes with an abnormal liver, the researchers write, then
they should ask whether the athletes are using potentially 'dangerous' supplements like whey protein and creatine.

We put ‘dangerous’ in quotes advisedly. We don’t actually believe that healthy people can get jaundice from creatine and
proteins. We agree with the opinion of a blogger – who by the way also works at Gaspari Nutrition – at bodybuilding.com
who suspects that the bodybuilder in question was also using steroids, and developed jaundice as a result of these.
[bodybuilding.com 2008/05/23]
It wouldn’t be the first time that bodybuilders have run into trouble for using hardcore doping materials, go to a doctor
and then don’t tell the whole truth about what they are using. In the past we have written about a bodybuilder who had
taken an overdose of DNP, got sick and went to hospital – where he died because he didn’t
dare to say what he had been using. In another article a bodybuilder almost lost a testicle because
at first he didn’t tell the doctors that he had been using Pregnyl.
There are plenty of reports in the medical literature of the side-effects supplements or low doses of doping drugs, which
should be enough to make the readers’ hair stand on end. But these reports are probably the result of silent use of
forbidden substances – and in high quantities. For example, there was the bodybuilder who said he had developed gyno from
using Tribulus terrestris supplements.
[Breast. 2004 Oct;13(5):428-30.] Other bodybuilders have said
that they got liver damage from androstenedione and a blood clot the size of a brick from
taking deca and proviron.
[N Engl J Med. 1999 Apr 8;340(14):1123-4.]
Sources:
Semin Liver Dis. 2008 May;28(2):226-31.
Supplement protects steroids users’ livers
A preparation made by Natterman, which supposedly protects the liver and which chemical athletes are using more and more
often: Greek researchers refer to it in their study as compound N. The researchers, at Thessaly University, set up an
experiment to determine whether compound N really works.

|
After a bit of surfing we worked out that Compound N is Essentiale forte.
[Google]
A packet contains a couple of dozen capsules, each of which contains 300 mg of polyene phosphatidylcholine. This is
choline-phosphoric acid with two unsaturated fatty acids attached, usually linoleic acid. It probably resembles the
phosphatidylcholine found in soya.
In addition, each capsule contains 6 mg of vitamin B1, 6 mg of vitamin B2, 6 mg of vitamin B6, 6 mcg of vitamin B12,
30 mg of nicotinamide and 6 mg of vitamin E.
The Greeks, who by the way have also done research on the psychological effects of anabolic steroids, did an experiment
with three hundred and twenty athletes. Half of them, one hundred and sixty athletes, used steroids. Of these chemical
athletes, the researchers gave forty Essentiale forte: two capsules a day taken with food.
All of the chemical athletes took steroids and the researchers monitored them for eight weeks. The list below gives you
an idea of what the Greek bodybuilders were using.
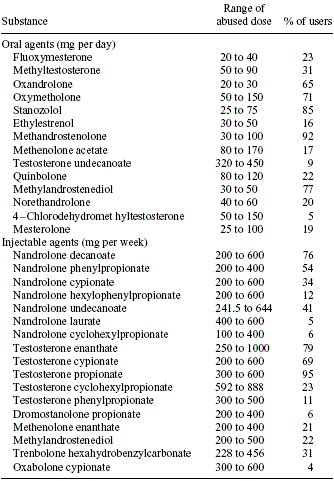
Yes, the list puzzled us too. Quinbolone? [An enol-ether of boldenone – Ed.] Oxabolone? [Nandrolone with a hydroxyl
group on C4 – Ed.] Are these products still on the market? And where are the 'new' designer steroids? Surely athletes
in Greece use products like 1-Test and Madol too?
Still.
During the eight-week period the researchers measured the athletes’ concentrations of the following enzymes: aspartate
aminotransferase (AST/SGOT), alanine aminotransferase (ALT/SGPT), lactate dehydrogenase (LDH), alkaline phosphatase (ALP),
gamma-glutamyltranspeptidase (gamma-GT) and creatine kinase (CK).
The more enzymes in the blood, the harder a time the liver is having.
The figure below shows what happened to the liver enzyme concentrations. Group A = chemical athletes who took
Essentiale forte as well. Group B = chemical athletes who did not take a liver-protection supplement. Group C =
‘natural’ athletes.

The researchers are not sure how phosphatidylcholine and vitamins protect steroids users’ livers. They suspect that
the mixture strengthens the membranes of the liver cells. The livers of steroids users have to work hard to break down
all the extra substances they are subjected to. And as a result the liver cells ‘cut down’ on metabolising fats. The
liver gets fattier because the liver cells are no longer burning fat well [beta-oxidation – Ed.] and maintenance of
the cell membranes made up of fatty acid chains gets neglected. The supplement helps the liver cells to perform these
functions.
Sounds attractive, but to be honest we find it difficult to believe that simply taking vitamin B pills and lecithin
capsules can protect steroids users’ livers. And we’re probably not the only ones who are sceptical. Reading between
the lines you can also see that the Greeks had trouble getting their study published.
That’s why they use long-winded sentences like: "the results from our cohort of similarly exercising individuals suggest
that polyunsaturated phospholipids in combination with vitamins of the B complex protect hepatic cells from AAS-induced
damage." According to the Greeks Compound N is not a supplement but "a controlled pharmaceutical agent".
But still. It’s not totally impossible that the Greeks have discovered something that will enable thousands of steroids
users to juice more safely.
More damaged livers from superdrol and madol
Liver specialists at the Henry Ford Hospital in Detroit have reported another three cases of bodybuilders who
developed liver damage as a result of using designer supplements. The men used Anabolic Xtreme’s
Superdrol or BMF Hardcore’s M-Test 2, a product containing the steroid
madol.
The Superdrol user was 21. He was nauseous, had stopped eating, had jaundice and itched all over. He’d been using

|
When the symptoms had got worse after two weeks, the doctors gave him prednisone. The anti-inflammatory worked and
after another six weeks, he'd made a pretty good recovery.
Superdrol is a steroid whose structure and synthesis resemble those of
oxymetholone. It was also developed and tested
in the late fifties by the makers of oxymetholone, the American pharmaceutical company Syntex. Although superdrol
[see structural formula below] looked like a promising anabolic steroid in animal tests, in subsequent tests potential
side effects showed up.

|
The effects of this move are clearly seen in the medical journals. In 2006 doctors in Phoenix, Arizona published an
article on the case of bodybuilder who became fatally ill after using superdrol.
[Am J Gastroenterol. 2006 Nov;101(11):2659-62.]
A few months later doctors at Johns Hopkins University published another case and a case in which another designer steroid
– Halodrol – had caused liver damage.
[Clin Gastroenterol Hepatol. 2007 Jul;5(7):809-12.]
And another few months after that, doctors from Burlington wrote about five more bodybuilders who had developed
liver problems as a result of superdrol. [Clin Gastroenterol Hepatol. 2008 Feb;6(2):255-8.]
What patient 2 used was not clear, but it contained at least DHEA. Of the three men referred to in the study, number 2
got off the most lightly. He recovered spontaneously a couple of weeks after he had stopped using the supplement.
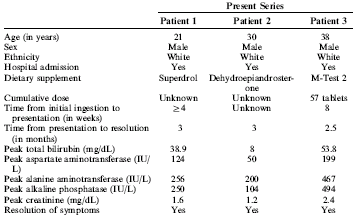
Patient 3 used M-Test 2, a designer supplement containing the steroid madol. [Structural formula shown below.] Madol was
rediscovered by Patrick Arnold, who produced it as an invisible steroid for Balco, but it was picked up later by designer
supplement manufacturers. Madol was also developed by Syntex, a manufacturer that carried out promising animal tests on it
in the sixties.

|
Not much is known about the side-effects of madol. It is not carcinogenic, say German researchers. But they did discover that
it enlarged the heart muscle in animal tests. The enlargement itself was not dangerous, but the researchers were not entirely
convinced. In the bodybuilding circuit however there are few stories around of users who have developed liver problems as a
result of using madol.
The sick madol user had to be given prednisone in the end, after which he recovered.
The doctors did not test the preparation the patient had used. And we
wonder quite honestly if it only contained madol. According
to the study, the bodybuilder became ill after he had taken 57 capsules
over a period of a few weeks. That would be impossible with

|
BMF Hardcore, the maker of M-Test, has heavier oral designer supplements
among its products. The Canadian government has issued
warnings about these. Maybe something went wrong during the production
of M-Test 2. Or perhaps the doctors got it wrong that Patient
3 used the BMF product.
The doctors also read a couple of articles written by colleagues and
summarized the information they found. This resulted in the
table you see here below.
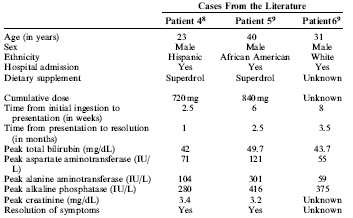
Liver specialists should get extra training on designer supplements that
contain oral anabolic steroids, the article concludes.
"The rapid reporting of several cases of AAS-induced liver injury from
dietary supplements emphasizes the growing emergence and
importance of this condition and the need for clinicians to become aware
of the sequelae of jaundice and renal failure, especially
among young men who are unknowingly consuming hepatotoxic agents."
=============================
Acute liver injury induced by weight-loss herbal supplements
World J Hepatol. 2010 November 27; 2(11): 410–415.
NCBI The National Center for Biotechnology Information advances science and health by providing access to biomedical and genomic information.
Published online 2010 November 27. doi: 10.4254/wjh.v2.i11.410
PMCID: PMC3004035
Abstract
We
report three cases of patients with acute liver injury induced by
weight-loss herbal supplements. One patient took Hydroxycut while the
other two took Herbalife supplements. Liver biopsies for all patients
demonstrated findings consistent with drug-induced acute liver injury.
To our knowledge, we are the first institute to report acute liver
injury from both of these two types of weight-loss herbal supplements
together as a case series. The series emphasizes the importance of
taking a cautious approach when consuming herbal supplements for the
purpose of weight loss.
Keywords: Hydroxycut, Herbalife, Hepatotoxicity, Herbal, Weight-loss
INTRODUCTION
We have seen a significant increase in the popularity and usage of over the counter herbal supplements over the past few years[1].
Unfortunately, the majority of these herbal supplements are not
regulated by drug administrations worldwide. Many herbal supplements
contain compounds that carry potentially severe side effects including
hepatotoxicity. We report three cases of acute liver injury induced by
weight-loss herbal supplements. Hydroxycut (MuscleTech, Mississauga,
Ontario, Canada) (case 1) and Herbalife (Herbalife, Los Angeles, USA)
(cases 2 and 3) supplements were the suspected culprits of acute liver
injury. Hydroxycut is a popular dietary supplement consisting of a
variety of herbal mixtures that claims to enhance the weight loss
process[2].
Acute liver injury associated with Hydroxycut use has been previously
reported, but only one case had liver biopsy data showing cholestasis
and portal inflammation[3-6].
Similarly, Herbalife weight-loss dietary products are popular
supplements consisting of a variety of herbal mixtures that claim to
facilitate weight reduction[7].
Cases of acute liver injury after consumption of Herbalife products
have been previously reported, with two patients developing fulminant
liver failure requiring liver transplantation. The first patient
survived while the second died[8-11]. In all of our cases, we were able to demonstrate drug-induced acute liver injury on liver biopsy specimens.
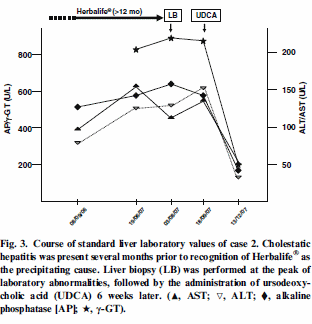
CASE REPORT
Case 1
A
31-year-old woman presented to our hospital complaining of 2-wk history
of fatigue, jaundice, and nausea. She denied any prior medical or
surgical conditions, family history of liver disease, and acetaminophen
or prescription medication use. She further denied history of blood
transfusion, tattoo, alcohol use, or recreational drug use. She had been
taking Hydroxycut for one year to enhance her weight loss. She had been
taking the recommended dose of 2 tablets twice a day.
The
patient was afebrile with normal hemodynamics upon presentation. Her
physical examination was remarkable for generalized jaundice, scleral
icterus, and mild upper quadrant tenderness to palpation without rebound
or guarding. Initial laboratory studies were significant for serum
aspartate aminotransferase (AST) level of 1407 U/L (normal range 15-41),
serum alanine aminotransferase (ALT) level of 1278 U/L (normal range
7-35), serum alkaline phosphatase of 256 U/L (normal range 38-126),
serum total bilirubin (TB) of 7.1 mg/dL (normal range 0.2-1.2), and
international normalized ratio (INR) of 1.3 I/U (normal range 0.8-1.2).
Given these findings, patient was admitted to the hospital for a higher
level of care.
Standard blood tests were
negative for hepatitis A, B, C, E, Ebstein Barr virus (EBV),
cytomegalovirus (CMV), human immunodeficiency virus (HIV), antinuclear
antibody, anti-smooth muscle antibody, anti-liver/kidney microsomal
antibody, alpha-1-antitrypsin deficiency, and anti-mitochondrial
antibody. Serum acetaminophen and urine toxicity screens were negative.
Serum ceruloplasmin, ferritin, iron studies, and immunoglobulins were
all within the normal range. Right upper quadrant ultrasound showed
diffuse echogenicity of the liver. Liver biopsy was performed and showed
multi-lobular necrosis consistent with acute toxic necrosis and
fulminant hepatitis (Figure (Figure11).

Liver
biopsy showed extensive patchy areas of multilobular necrosis with only
bile ducts remaining, extensive ductal metaplasia, severe lymphocytic
and macrophages infiltration of portal tracts and lobular parenchyma and
patchy plasma cell infiltrates. ...
The
patient’s liver function tests peaked 4 d after admission with serum
AST level of 1613 U/L, ALT level of 1227 U/L, serum alkaline phosphatase
of 268 U/L, serum TB of 10.5 mg/dL, and INR staying at 1.3 I/U. She did
not develop evidence of hypoglycemia or portal-systemic encephalopathy.
Her jaundice and scleral icterus resolved over the following 2-wk. Her
liver tests gradually improved within the following few months.
Case 2
A
37-year-old woman presented to our hospital with a 1-mo history of
diffuse abdominal pain, mild nausea, and painless jaundice. She denied
any past medical or surgical history, family history of liver disease,
or any alcohol or illicit substance abuse. She admitted that she had
been taking Herbalife dietary supplements for the past 3-mo in an
attempt to lose weight. Her Herbalife regimen consisted of the Formula
One Nutritional Shake Mix, the Multivitamin Complex, the Cell Activator,
the Cell-U-Loss, the Herbal Concentrate Original, and the Total Control
formula.
The patient was afebrile with normal
vital signs on presentation. Her physical exam was noticeable for
bilateral scleral icterus and generalized jaundice. Her abdominal exam
revealed a non-tender, non-distended abdomen with no stigmata of liver
disease. Initial laboratory studies were significant for an AST level of
2199 U/L, serum ALT level of 2068 U/L, serum alkaline phosphatase of
185 U/L, and TB of 15.3 mg/dL. All other laboratory values, including
amylase, lipase, and INR, were within normal limits. Given these lab
abnormalities, the patient was admitted to the hospital for further
work-up.
Standard blood tests were negative for
hepatitis A, B, C, E, EBV, CMV, HIV, antinuclear antibody, anti-smooth
muscle antibody, anti-liver/kidney microsomal antibody,
alpha-1-antitrypsin deficiency, and anti-mitochondrial antibody. Serum
acetaminophen and urine toxicity screens were negative. Serum
ceruloplasmin, ferritin, iron studies, and immunoglobulins were all
within normal range. A computerized tomography (CT) scan of the abdomen
and pelvis with intravenous (IV) contrast showed multiple low-density
lesions in the liver measuring up to 8-mm. A liver biopsy revealed acute
necrotizing hepatitis both centrolobular and periportal, consistent
with a drug-induced etiology (Figure (Figure2).2).
However, her liver biopsy specimens also showed evidence of bridging
fibrosis, which suggest some degree of chronic liver disease but with
drug-induced injury in addition.

Liver
biopsy was performed and showed periportal bridging fibrosis, ductal
metaplasia, cholestasis, moderate intralobular lymphocytic infiltration,
and troxis necrosis and apoptosis consistent with drug-induced
hepatitis on top chronic liver disease. ...
The
patient was treated supportively with fluids and nutrition. Her liver
tests steadily declined from the day of admission and on hospital day 8
(day of discharge) her liver tests revealed a AST level of 1788 U/L, ALT
level of 1501 U/L, and serum alkaline phosphatase of 183 U/L. The only
laboratory value to increase was the patient's serum TB, which was at
29.9 mg/dL on discharge. The patient did not develop encephalopathy,
hypoglycemia, or any other complications. The patient was followed for
several months, throughout which her symptoms continued to improve.
At
her 2-mo follow-up, the patient's icterus and jaundice had resolved
completely. Her labs at this time showed a serum AST level of 51 U/L,
serum ALT level of 43 U/L, serum alkaline phosphatase of 65 U/L, and
serum TB of 1.1 mg/dL.
Case 3
A
53-year-old previously healthy woman presented with a 3-wk history of
painless jaundice and pruritus. She denied any family history of liver
disease, or any alcohol or illicit substance abuse. She had not been
taking any new prescribed medications. On further questioning about
over-the-counter supplements she divulged a 4-mo history of consuming
various Herbalife weight loss products in the form of shakes, teas and
pills.
On physical exam the patient’s vital
signs were within normal limits. On general inspection she had scleral
icterus and jaundice, with evidence of excoriations. A 2-cm palpable
liver edge could be appreciated, that was tender to touch. There were no
other signs of chronic liver disease. Initial laboratory values
revealed a hepatocellular pattern of injury, with an AST of 1282 U/L,
ALT of 983 U/L, and alkaline phosphatase of 292 U/L, with a TB of 18.2
mg/dL. An ultrasound showed borderline hepatomegaly of 17-cm.
Standard
blood tests for hepatitis A, B, C, E, EBV, CMV, HIV, antinuclear
antibody, anti-smooth muscle antibody, anti-liver/kidney microsomal
antibody, alpha-1-antitrypsin deficiency, and anti-mitochondrial
antibody were negative. Serum acetaminophen and urine toxicity screens
were negative. Serum ceruloplasmin, ferritin, iron studies, and
immunoglobulins were all within normal range.
Liver biopsy was performed and showed cholestasis, consistent with drug induced hepatitis (Figure (Figure3).3). 2-mo after complete abstinence from the Herbalife supplements her jaundice resolved, as did her liver tests.
DISCUSSION
Acute
liver injury induced by over the counter weight-loss herbal supplement
Hydroxycut and Herbalife products have been reported previously[3-6,8-11].
These case reports were limited by the fact that liver biopsies were
performed in only a few patients, confirming clinical suspicions
histologically. In terms of our patients, all three had liver biopsy
performed and all showed some common morphological features including
diffuse lymphocytic infiltration of sinusoids and portal tracts, ductal
metaplasia and toxic necrosis. Some variations of morphological features
could be explained by predominance of intrinsic or idiosyncratic
mechanisms of hepatic injury, individual patient response to the
affecting drug and duration of injury. The patients’ liver biopsy
specimens were stained with periodic acid-Schiff (PAS) stain with
diastase. No hyaline globules were identified in any of the three cases.
The absence of histological findings and the fact that our patients had
no history of chronic obstructive pulmonary disease excluded diagnosis
of alpha-1-antitripsin deficiency in all three cases. Prussian blue and
copper stains did not reveal excessive iron or copper depositions in the
hepatocytes and Kupffer cells.
Only one
previous case of Hydroxycut-induced acute liver injury had reported
findings on liver biopsy. Although the most likely explanation for the
mechanism of liver injury caused by these herbal products is
idiosyncratic reaction, one of the ingredients in Hydroxycut, green tea
extract (Camellia sinensis), has been linked with acute liver injury in other over the counter weight-loss herbal supplements[12-20]. In fact, the weight-loss herbal supplement Exolise (Arkophama, Carros, France), which also contained C. sinensis, was withdrawn from the market because it was linked to multiple cases of liver injury[13].
Furthermore, several cases of hepatotoxicity were associated with
another herbal weight-loss supplement, Cuur (Scandinavian Clinical
Nutrition, Sweden), which also contains the ethanolic dry extract of
green tea (C. sinensis)[15]. Rechallenging patients with the same product led to hepatotoxicity, confirming the role of C. sinensis[12,16].
In all reported cases of acute liver injury induced by Hydroxycut,
patients’ liver function tests recovered over time following cessation
of the product. However, there have been cases of liver failure caused
by green tea extract C. sinensis, requiring orthotopic liver transplantation[13,16].
The
liver biopsy obtained in our patient who took Hydroxycut showed
multi-lobular necrosis consistent with acute toxic necrosis and
fulminant hepatitis. These findings are similar to the findings in
patients with liver injury associated with green tea extract C. sinensis, where prominent necrosis with inflammatory reaction is the hallmark presentation[15,16].
The exact mechanism of hepatotoxicity induced by Hydroxycut is unknown. However, as this product contains green tea extract C. sinensis,
it is possible that this may play a role in acute liver injury caused
by Hydroxycut. Prior investigation into the mechanism of hepatotoxicity
by green tea extract was inconclusive[21].
Others have hypothesized that a possible allergic reaction to the green
tea extract, contamination during the production of the extract or a
metabolic idiosyncrasy are possible mechanisms of liver injury in these
patients[16].
Both
of our patients took several Herbalife weight-loss herbal products
concurrently, similar to most of the previously reported cases of
hepatotoxicity due to Herbalife products[8-11].
Therefore, it is difficult to identify the exact ingredient or
mechanism that causes the liver injury, as in the previously documented
cases[8-11]. In a previously reported case, one investigator was able to isolate contamination with Bacillus subtilis, in which the bacterial supernatant caused dose-dependent increase of LDH leakage in HepG2 cells[8]. Although not commonly known as a human pathogen, B. subtiliis has been reported to cause food poisonings and a case of cholangitis in an immunocompromised patient[22-23].
Investigators have also suggested that another explanation for
hepatotoxicity due to Herbalife products could be secondary to locally
restricted contamination with chemicals such as softeners,
preservatives, flavor enhancers, pesticides, or heavy metals either
intentionally added during the production process or contained in the
unrefined raw herb extracts[24].
To date, Herbalife has refused to provide detailed analyses of their products’ composition and ingredients[25].
This contamination hypothesis could also explain the different patterns
of pathology seen on liver biopsy specimens previously observed in
patients with hepatotoxicity from Herbalife products as both
predominantly cholestatic injury pattern and acute hepatitis pattern
have been reported[8-11]. Our patients had findings consistent with acute hepatitis due to drug-induced liver injury on their liver biopsy specimens.
Due
to the obesity epidemic, the usage of weight-loss herbal supplements
has flourished. Green tea extract is one of the key components in many
of the over-the-counter weight-loss herbal supplements. Although
significant liver injury induced by herbal supplements taken for weight
loss purposes is a rare event, we cannot ignore the fact that there have
been multiple reported cases in the medical literature of
hepatotoxicity associated with weight-loss herbal supplements including
Hydroxycut and Herbalife products. Even though our patients successfully
recovered from the adverse reactions, we must bear in mind that the
hospitalization and medical care of these patients were associated with
significant cost and healthcare resource utilization, while there is no
evidence that herbal supplements can help with weight-loss[26].
We must also consider the impact on patients with underlying chronic
liver disease, in whom herbal weight loss medications could cause
worsening in their synthetic function and even fulminant failure. In May
of 2009, the US Food and Drug Administration warned consumers to
immediately stop using Hydroxycut products, citing linkage to liver
damage in one patient who died due to liver failure[27].
However, Hydroxycut products are currently still available in many
parts of the world. Likewise, Herbalife products are widely available
globally. Therefore, it is these authors’ view that closer monitoring of
patients taking weight-loss herbal supplements as well as tighter
regulation from government drug agencies is warranted. Furthermore, our
cases once again demonstrated the importance of questioning patients
regarding the usage of herbal or nutritional supplements at the time of
evaluation.
Footnotes
Supported by Harbor UCLA Medical Center and The Division of Gastroenterology
Peer
reviewers: Mary Ko Manibusan, Co-Chair, US Environmental Protection
Agency, Office of Pesticide Programs, Health Effects Division - Crystal
City, 8136 Viola Street, Springfield, VA 22152, United States; Ferruccio
Bonino, Professor, Chief, Scientific Officer, Fondazione IRCCS Ospedale
Maggiore Policlinico Mangiagalli e Regina Elena, Via F. Sforza 28,
Milano 20122, Italy
S- Editor Zhang HN L- Editor Hughes D E- Editor Liu N
References
1. Kelly
JP, Kaufman DW, Kelley K, Rosenberg L, Anderson TE, Mitchell AA. Recent
trends in use of herbal and other natural products. Arch Intern Med. 2005;165:281–286. [PubMed]
2. Available from: http://www.hydroxycut.com/products/hydroxycut/index.shtml.
3. Stevens
T, Qadri A, Zein NN. Two patients with acute liver injury associated
with use of the herbal weight-loss supplement hydroxycut. Ann Intern Med. 2005;142:477–478. [PubMed]
4. Jones FJ, Andrews AH. Acute liver injury associated with the herbal supplement hydroxycut in a soldier deployed to Iraq. Am J Gastroenterol. 2007;102:2357–2358. [PubMed]
5. Shim M, Saab S. Severe hepatotoxicity due to Hydroxycut: a case report. Dig Dis Sci. 2009;54:406–4088. [PubMed]
6. Dara
L, Hewett J, Lim JK. Hydroxycut hepatotoxicity: a case series and
review of liver toxicity from herbal weight loss supplements. World J Gastroenterol. 2008;14:6999–7004. [PMC free article] [PubMed]
7. Available from: http://products.herbalife.com/weight-management.
8. Stickel
F, Droz S, Patsenker E, Bögli-Stuber K, Aebi B, Leib SL. Severe
hepatotoxicity following ingestion of Herbalife nutritional supplements
contaminated with Bacillus subtilis. J Hepatol. 2009;50:111–117. [PubMed]
9. Elinav
E, Pinsker G, Safadi R, Pappo O, Bromberg M, Anis E, Keinan-Boker L,
Broide E, Ackerman Z, Kaluski DN, et al. Association between
consumption of Herbalife nutritional supplements and acute
hepatotoxicity. J Hepatol. 2007;47:514–520. [PubMed]
10. Schoepfer
AM, Engel A, Fattinger K, Marbet UA, Criblez D, Reichen J, Zimmermann
A, Oneta CM. Herbal does not mean innocuous: ten cases of severe
hepatotoxicity associated with dietary supplements from Herbalife
products. J Hepatol. 2007;47:521–526. [PubMed]
11. Duque JM, Ferreiro J, Salgueiro E. Hepatotoxicity associated with the consumption of herbal slimming products. Med Clin (Barc) 2007;128:238–239. [PubMed]
12. Bonkovsky HL. Hepatotoxicity associated with supplements containing Chinese green tea (Camellia sinensis) Ann Intern Med. 2006;144:68–71. [PubMed]
13. Gloro
R, Hourmand-Ollivier I, Mosquet B, Mosquet L, Rousselot P, Salamé E,
Piquet MA, Dao T. Fulminant hepatitis during self-medication with
hydroalcoholic extract of green tea. Eur J Gastroenterol Hepatol. 2005;17:1135–1137. [PubMed]
14. Jimenez-Saenz
M, Martinez-Sanchez C. Green tea extracts and acute liver failure: the
need for caution in their use and diagnostic assessment. Liver Transpl. 2007;13:1067. [PubMed]
15. Bjornsson E, Olsson R. Serious adverse liver reactions associated with herbal weight-loss supplements. J Hepatol. 2007;47:295–297; author reply 297-298. [PubMed]
16. Molinari
M, Watt KD, Kruszyna T, Nelson R, Walsh M, Huang WY, Nashan B,
Peltekian K. Acute liver failure induced by green tea extracts: case
report and review of the literature. Liver Transpl. 2006;12:1892–1895. [PubMed]
17. Thiolet C, Mennecier D, Bredin C, Moulin O, Rimlinger H, Nizou C, Vergeau B, Farret O. [Acute cytolysis induced by Chinese tea] Gastroenterol Clin Biol. 2002;26:939–940. [PubMed]
18. Vial
T, Bernard G, Lewden B, Dumortier J, Descotes J. Acute hepatitis due to
Exolise, a Camellia sinensis dried ethanolic extract [letter] Med Clin (Barc) 2003;121:598–599. [PubMed]
19. García-Morán
S, Sáez-Royuela F, Gento E, López Morante A, Arias L. Acute hepatitis
associated with Camellia thea and Orthosiphon stamineus ingestion
[letter] Gastroenterol Hepatol. 2004;27:559–560. [PubMed]
20. Porcel JM, Bielsa S, Madronero AB. Hepatotoxicity associated with green tea extracts [electronic letter]. Accessed by www.annals.org on June 3. 2005.
21. Schmidt
M, Schmitz HJ, Baumgart A, Guédon D, Netsch MI, Kreuter MH, Schmidlin
CB, Schrenk D. Toxicity of green tea extracts and their constituents in
rat hepatocytes in primary culture. Food Chem Toxicol. 2005;43:307–314. [PubMed]
22. Kramer JM, Gilbert RJ. Bacillus cereus and other Bacillus species. In: Doyle MP, editor. Foodborne bacterial pathogens. New York: Decker; 1989. pp. 21–70.
23. Wallet
F, Crunelle V, Roussel-Delvallez M, Fruchart A, Saunier P, Courcol RJ.
Bacillus subtilis as a cause of cholangitis in polycystic kidney and
liver disease. Am J Gastroenterol. 1996;91:1477–1478. [PubMed]
24. De Smet PA. Herbal remedies. N Engl J Med. 2002;347:2046–2056. [PubMed]
25. Stickel F. Slimming at all costs: Herbalife-induced liver injury. J Hepatol. 2007;47:444–446. [PubMed]
26. Lenz TL, Hamilton WR. Supplemental products used for weight loss. J Am Pharm Assoc (2003) 2004;44:59–67; quiz 67-68. [PubMed]
27. Available from: http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm152152.htm.
==================================
Ha Na Yang,1 Dong Joon Kim,



===========================
===========================
SUITE101

==================================
Aloe-induced Toxic Hepatitis
J Korean Med Sci. 2010 March; 25(3): 492–495.
Published online 2010 February 17. doi: 10.3346/jkms.2010.25.3.492
PMCID: PMC2826749
Ha Na Yang,1 Dong Joon Kim, 1 Young Mook Kim,1 Byoung Ho Kim,1 Kyoung Min Sohn,1 Myung Jin Choi,1 and Young Hee Choi2
1 Young Mook Kim,1 Byoung Ho Kim,1 Kyoung Min Sohn,1 Myung Jin Choi,1 and Young Hee Choi2
This article has been cited by other articles in PMC.
Abstract
Aloe
has been widely used in phytomedicine. Phytomedicine describes aloe as a
herb which has anti-inflammatory, anti-proliferative, anti-aging
effects. In recent years several cases of aloe-induced hepatotoxicity
were reported. But its pharmacokinetics and toxicity are poorly
described in the literature. Here we report three cases with
aloe-induced toxic hepatitis. A 57-yr-old woman, a 62-yr-old woman and a
55-yr-old woman were admitted to the hospital for acute hepatitis. They
had taken aloe preparation for months. Their clinical manifestation,
laboratory findings and histologic findings met diagnostic criteria
(RUCAM scale) of toxic hepatitis. Upon discontinuation of the oral aloe
preparations, liver enzymes returned to normal level. Aloe should be
considered as a causative agent in hepatotoxicity.
Keywords: Aloe, Hepatitis, Toxic
INTRODUCTION
The
demand for dietary supplements has continually increased in recent
years as the concept of 'well being' widely spread in Korea. The market
value for dietary supplements in Korea was approximately 600 billion won
(600 million USD) in year 2005 (1), and personal spending was approximately 950,000 won (950 USD) per year in 2005 (2). One of the leading products in Korea's dietary supplements market is aloe.
Aloe
has been purported to have positive effects on wound healing, recovery
from burn injury, cell growth, and immune modulation. However, cases of
aloe-induced toxic hepatitis have been reported since 2005. In
particular, there have been one each in Germany (3), Turkey (4), and USA (5). In Switzerland (6),
10 cases of hepatotoxicity associated with dietary supplements from
Herbalife® products were reported. Although 2 of those 10 cases took
aloe preparation, the causality assessment between aloe and toxic
hepatitis could not be done due to multiple Herbalife® products.
We report 3 cases of aloe-induced toxic hepatitis in Korea.
CASE REPORTS
Case 1
A
57-yr-old female patient with a 2 month history of dyspepsia was
presented to our department. Past medical history and family history did
not reveal any significant disease. She also used drugs for arthralgia
intermittently for several years. She did not take any alcohol. She had
taken aloe tablets containing 250 mg of an extract of Aloe arborescens and 28.5 mg of an extract of Aloe vera (Fig. 1) for about 6 months prior to the admission. On admission, the patient's physical examination was normal.

Preparation of aloe which the patient had taken. (A) Container bottle and tablets. (B) Packs of aloe extract.
Laboratory
abnormalities included aspartate aminotransferase (AST) 331 IU/L,
alanine aminotransferase (ALT) 565 IU/L, alkaline phosphatase (AP) 309
IU/L. Anti-HAV IgM, anti-HCV, HCV PCR, and anti-HEV IgM were negative.
Anti-HBs IgG was positive. There was no serologic evidence for recent
infections with cytomegalovirus (CMV), Epstein-Barr-virus (EBV), or
herpes simplex virus (HSV). Autoimmune markers were all negative.
Abdominal ultrasonography showed reduced echogenicity of liver.
Dilatation of intra- or extrahepatic bile ducts was absent. Liver biopsy
revealed moderate portal infiltrates consisting of eosinophilis,
neutrophils, and monocytes. There were inflammatory cell infiltration
and acidophil body on the hepatic lobule. There was no bile stasis (Fig. 2).

Histopathological findings of the liver. (A) The portal area and lobular area show inflammatory cell infiltration (H&E, ×100). (B) The acidophilic body and ballooning cell change are noted (H&E, ×200).
Aloe
tablets was immediately discontinued. ALT was highest (926 IU/L) on the
12th day of admission and gradually decreased to 452 IU/L on the 25th
day of admission. ALT as well as total bilirubin gradually returned to
normal level over several weeks (Fig. 3).
Using the Roussel Uclaf Causality Assessment Method for determining
drug hepatotoxicity (RUCAM) scale, this case was scored as 'probable' (Table 1).
The type of liver injury was determined as 'hepatocellular' since R
ratio (serum activity of ALT/serum activity of AP) was 10 (7).
Case 2
A
62-yr-old female patient was presented to our department with a week
history of fatigue. Past medical history and family history did not
reveal any significant disease. The patient did not take any alcohol or
durgs. She had taken aloe powder containing 420 mg of an extract of Aloe vera (Fig. 1B)
for about 3 months prior to the admission. Physical examination
revealed jaundice on her sclera. She was the sales person of the aloe
product she was taking.
Laboratory abnormalities
included AST 1,477 IU/L, ALT 1,564 IU/L, total bilirubin 14.64 mg/dL.
Anti-HAV IgM, anti-HCV, HCV PCR, and anti-HEV IgM were negative.
Anti-HBs IgG was positive. IgM antibody for HSV was in trace, IgM
antibody for CMV was negative. Abdominal ultrasonography was normal.
Liver biopsy revealed severe portal and lobular infiltrates consisting
of neutrophils and monocytes. There were several acidophilic bodies and
ballooning cell change in hepatic lobule. There were bile-stasis and
bile stained Kupffer cells (Fig. 2B).
Aloe
extract was immediately discontinued. ALT gradually decreased to 452
IU/L which was lower than half of the peak value on the 17th day of
admission. When she was discharged ALT and AST were normal. We explained
to her about the aloe-induced hepatotoxity and advised not to take it
anymore. However, the patient started taking the same aloe extract again
1 month after her discharge from the hospital. A month later, follow-up
liver function test showed AST 477 IU/L, ALT 785 IU/L, and AP 165 IU/L (Fig. 4).
Since a hepatitis recurred after re-challenge of aloe extract, we could
confirm her diagnosis as aloe-induced toxic hepatitis. The RUCAM scale
was scored as 'definite' (Table 1). The type of liver injury was determined as 'hepatocellular' since R ratio was 39.

Case
2. Upon discontinuation of the oral aloe preparation, liver enzymes
returned to normal level. After re-challenge (arrow), liver enzymes go
up again.
Six months later, the
patient was presented to our department with a 2 week history of
jaundice. Laboratory test showed AST 752 IU/L, ALT 1,135 IU/L, AP 243
IU/L and total bilirubin 15.81 mg/dL. We recommended admission, but she
refused to be admitted and she never visited our hospital again.
Case 3
A
55-yr-old female patient was presented to our department with a 3 month
history of epigastric discomfort. Past medical history and family
history did not reveal any significant disease. The patient did not take
any alcohol or drugs. She had taken aloe extracts (Fig. 1)
for about 5 months prior to the admission. On admission, the patient's
physical examination was normal except mild tenderness on epigastric
area.
Laboratory abnormalities included AST 344 IU/L,
ALT 666 IU/L, AP 298 IU/L. Anti-HAV IgM, anti-HCV, HCV PCR, and anti-HEV
IgM were negative. Anti-HBs IgG was positive. Autoimmune markers were
negative. Abdominal ultrasonography showed increased echogenicity of
liver. Dilatation of intra- or extrahepatic bile ducts was absent.
Aloe
extract was immediately discontinued. ALT was highest (666 IU/L) on the
2nd day of admission, and gradually decreased. ALT was 484 IU/L on her
discharge (on the 5th day of admission). After 4 days, she visited our
department for follow-up. Liver function test was normal at that time
(AST 29 IU/L, ALT 11 IU/L, AP 190 IU/L) (Fig. 5). The RUCAM scale was scored as 'probable' (Table 1). The type of liver injury was determined as 'hepatocellular' since R ratio was 11.
DISCUSSION
The
major driving force for the growth of the dietary supplements market is
the perception that 'they are safe because they are natural'. However,
the recently reported cases of hepatotoxicity induced by natural
substances (8) indicate that natural substances may not be entirely safe.
There are about 400 species of aloe. Among them, particularly aloe vera has been used in phytomedicine. There have been positive reports on aloe vera as anti-inflammatory, anticancer, analgesic, anti-aging as well as liver protective. But, clinical effectiveness of aloe vera was not sufficiently defined because there were no large and randomized studies (9). In 1994, Korea's National Institute of Safety Research conducted an experiment on the efficacy and toxicity of aloe (10). There was no difference of natural killer cell activity between the aloe vera
gel treated and control animals. To observe the toxicity of aloe gel,
rats were given the high dose aloe orally. Any adverse effects were not
detected in hematological test, serum biochemistry, and
histopathological examination.
There are no specific
tests or diagnostic criteria for herbinduced hepatic injury. Careful
history taking, laboratory finding, and histopathology are used to
diagnose it. The best way to determine causing agent is re-challenging.
But it is not ethical and not applicable. Instead, the RUCAM scale is
used (7, 11).
Since
patients usually do not regard dietary supplements as 'real' medicine,
they may fail to mention it when physicians query medication history.
Physicians should keep in mind that dietary supplements can be the cause
of hepatotoxicity when querying medication history, and should educate
the lay public.
There are three types of acute liver injury by drug or herb (12):
hepatocellular, cholestatic, and mixed type. Our cases are
characterized as hepatocellular; there is a predominant initial
elevation of the ALT level. There are two proposed pathogeneses of drug
induced liver disease (13):
direct toxicity and idiosyncratic mechanism. It is more likely that an
idiosyncratic immunological mechanism (hypersensitivity) is responsible
for the cases. A role for hypersensitivity is further supported by the
presence of eosinophilic granulocytes in the periportal fields seen in
the biopsy. Hypersensitivity to aloe has been described in humans (14), and the patch test or allergic skin test showed positive results (15).
Herb
induced liver injury is an important problem in clinical setting,
because it can be an etiology of undiagnosed acute hepatitis. However,
there are few available data about the incidence and clinical
manifestation of dietary supplements such as aloe. Our cases emphasize
that physicians should consider various dietary supplements as causative
agents for hepatotoxicity.
References
1. Food and Drug Statistical Yearbook. Korea Food and Drug Administration. 2006. Aug, [accessed 2008 0ct 27]. Available at: URL: http://www.kfda.go.kr/open_content/main/main.php.
2. Yoo
TW, Kim BI, Kim JB, Kim DJ, Kim JW, Baik SK, Kim KS, Cheon GJ. The
survey for the actual condition of drug medication and development of
health care cost associated with toxic liver injury in Korean: a
multicenter study for the detection and the development of nationwide
reporting system of toxic liver injury. Korean J Hepatol. 2007;13:34–43. [PubMed]
3. Rabe C, Musch A, Schirmacher P, Kruis W, Hoffmann R. Acute hepatitis induced by an aloe vera preparation: a case report. World J Gastroenterol. 2005;11:303–304. [PubMed]
4. Kanat O, Ozet A, Ataergin S. Aloe vera-induced acute toxic hepatitis in a healthy young man. Eur J Intern Med. 2006;17:589. [PubMed]
5. Bottenberg MM, Wall GC, Harvey RL, Habib S. Oral aloe vera-induced hepatitis. Ann Pharmacother. 2007;41:1740–1743. [PubMed]
6. Schoepfer
AM, Engel A, Fattinger K, Marbet UA, Criblez D, Reichen J, Zimmermann
A, Oneta CM. Herbal does not mean innocuous: ten cases of severe
hepatotoxicity associated with dietary supplements from herbalife
products. J Hepatol. 2007;47:521–526. [PubMed]
7. Benichou C. Criteria of drug-induced liver disorders. Report of an international consensus meeting. J Hepatol. 1990;11:272–276. [PubMed]
8. Stickel F, Patsenker E, Schuppan D. Herbal hepatotoxicity. J Hepatol. 2005;43:901–910. [PubMed]
9. Vogler BK, Ernst E. Aloe vera: a systematic review of its clinical effectiveness. Br J Gen Pract. 1999;49:823–828. [PMC free article] [PubMed]
10. Jang DD, Cho JC, Choi KS, Kil HI. Studies of the effectiveness and toxicity of aloe vera gel. The report of National Institute of Safety Research. 1994;7:225–233.
11. Navarro VJ, Senior JR. Drug-related hepatotoxicity. N Engl J Med. 2006;354:731–739. [PubMed]
12. Chae HB. Clinical features and diagnosis of drug induced liver injury. Korean J Hepatol. 2004;10(Suppl1):7–18.
13. Kang DY. Pathologic features of toxic and drug induced liver injury. Korean J Hepatol. 2004;10(Suppl1):19–29.
14. Morrow DM, Rapaport MJ, Strick RA. Hypersensitivity to aloe. Arch Dermatol. 1980;116:1064–1065. [PubMed]
15. Lee EG, Kwon SH, Kim SH, Myung SJ, Choi JW, Kim YJ, Ahn Y. A case of hypersensitivity associated with oral aloe agent. J Asthma Allergy Clin Immunol. 2003;23:833–836.
Revisiting acute liver injury associated with herbalife products
Abstract
In
the November 27, 2010 issue of the World Journal of Hepatology (WJH),
three case reports were published which involved patients who had
consumed various dietary supplements and conventional foods generally
marketed as weight loss products. The reference to Herbalife products as
contaminated and generally comparable to all dietary supplements or
weight loss products is not scientifically supported. The authors
provided an insufficient amount of information regarding patient
histories, concomitant medications and other compounds, dechallenge
results, and product specifications and usage. This information is
necessary to fully assess the association of Herbalife products in the
WJH case reports. Therefore, the article does not objectively support a
causal relationship between the reported cases of liver injury and
Herbalife products or ingredients.
Keywords: Herbalife, Liver, Hepatotoxicity, Weight loss products, Dietary supplements
TO THE EDITOR
In the November 27, 2010 issue of the World Journal of Hepatology (WJH),
three case reports were published which involved patients who had
consumed various dietary supplements and conventional foods generally
marketed as weight loss products. Case 1 involved a patient who did not
consume Herbalife products, while Cases 2 and 3 each reportedly consumed
various Herbalife products. Herbalife fundamentally disagrees with the
conclusions made by the authors with regard to any cause and effect
relationship related to the intake of Herbalife products. First,
Herbalife is not a single product and no unique suspect product or
ingredient has been implicated in this paper amongst the reported cases.
In addition, the authors arbitrarily compared cases involving the use
of a single product (Hydroxycut) with patients who consumed a group of
totally unrelated products produced by the company Herbalife. To bundle a
brand of products such as Herbalife with another company that sells
different products simply because they are all dietary supplements is
not valid. Finally, there are specific considerations, in regard to the
two patients who consumed Herbalife products, that would render many of
the observations and conclusions discussed by the authors as speculative
and unsubstantiated. The specific and factual points supporting these
views are further detailed below.
Case 2 describes a
37-year-old female who developed symptoms of abdominal pain, mild
nausea, and painless jaundice 1 mo prior to presenting at the hospital[1].
Several pertinent negatives were disclosed by the authors, including
autoimmune markers and viral serology. According to the authors, the
patient did not report any pre-existing medical conditions for which the
onset had preceded the use of Herbalife products. However, the
pathology assessment concluded that this patient’s biopsy result was
consistent with chronic liver disease, in which case Herbalife products
were thought to have had an additive effect. This opinion contradicts
repeated statements by the authors that acute liver injury in each case
report was due to the use of herbal weight loss products. In addition,
the etiology of the pre-existing condition was not identified by the
authors, and there was no discussion regarding the role of the condition
in the acute onset of her symptoms. Furthermore, the dosage and
frequency at which this patient consumed Herbalife products is unknown.
Finally, the inconsistency of the objective findings with the patient’s
reported medical history would suggest that further investigation is
warranted. This should include a review of the patient’s pre-existing
condition, potential use of medications prescribed for her condition,
other compounds she may have been consuming, and the status of her
health prior to the reported incident. In the absence of the
aforementioned data, the exclusion of possible differential diagnoses is
not well-supported.
Case 3 describes a 53-year-old
female who developed symptoms of painless jaundice and pruritus 3 wk
prior to presenting at the hospital[1].
This patient denied family history of liver disease, but no discussion
was provided regarding her own medical history, other than the fact that
she reportedly denied the use of alcohol and did not engage in “illicit
substance abuse”. The authors further stated that the patient had not
been prescribed any new medications, which implies that she may have
been taking other agents concomitantly. However, information regarding
the use of concomitant medications, or the conditions for which she may
have been receiving treatment, was not disclosed. Such information is
critical and should have been obtained through follow-up review of the
patient’s previous medical records. Without this information, it is
unknown whether concomitant medication(s) were withdrawn and/or
accounted for during the dechallenge process. The patient’s use of
Herbalife products was also not specified by product names and it is
unknown whether the dosage and frequency of consumption was adherent to
recommendations indicated on the product label(s). In addition to the
absence of the aforementioned pertinent patient data, there are various
refutable facts that remain in regard to the comments and conclusions
made by the authors.
In their WJH article, the
authors concluded that it was difficult to isolate a single ingredient
or mechanism associated with acute liver injury for either patient
consuming Herbalife products[1].
In an effort to discuss potential causative agents for the reported
conditions in these patients, the authors extraneously reference
previously published case reports involving Herbalife products,
including those of two consumers who reportedly developed hepatotoxicity
following exposure to Bacillus subtilis (B. subtilis)[2].
In
review of this reference, it has been noted that there were various
critical deficiencies in the scientific methodology used to isolate B. subtilis
in the Herbalife samples reported to have been contaminated. For
example, a dose dependent increase in LDH leakage in HepG2 cells was
observed in the experimental assay, but investigators did not present
any control data for their experiments, nor did they present any data
that suggested this assay is a valid proxy for liver injury in healthy
individuals “in vivo”. Neither patient reported symptoms consistent with classical B. subtilis
food poisoning and they did not report testing the product for the
detection of cerulide or any of the reported heat-stable toxins
associated with certain strains of B. subtilis. Furthermore, the investigators did not enumerate the levels of B. subtilis
in the products tested or report testing relevant specimens from the
patients for these organisms or their toxins. This was a crucial step
missing in the reported investigation as all previous documented reports
find that high levels of the organism must be consumed to cause
illness. Herbalife products, consumed by the patients described in the WJH article, to date show no evidence of B. subtilis contamination. B. subtilis
infections are relatively rare and seldom contracted through food
sources. This bacterium is actually ubiquitous in nature and generally
recognized as safe with a history of safe use in food, and is considered
to be safe for the production of enzymes or ingredients for use in
food[3]. There have been reported cases of B. subtilis-related
gastroenteritis and other complications, usually involving
immuno-compromised patients or those with other underlying chronic
illnesses, which did not appear to be the case for any of the patients
presented in the WJH article. Therefore, it is highly unlikely that B. subtilis
could be the cause or have contributed to the severe hepatotoxicity of
patients in either the referenced article or the two patients discussed
in the WJH article.
The WJH authors
also suggest intentional or incidental contamination of Herbalife
ingredients and identify various potential sources, including unrefined
raw herbal extracts, heavy metals, pesticides, and additives[1].
However, some of the additives mentioned as potential contaminants by
the authors (e.g., flavoring, colors, and preservatives) are commonly
used and well-documented industry-wide as safe for consumption in
conventional foods, as well as dietary supplements,. In addition,
authors also reference an article from 2002 that reviews possible
contamination sources inherent to herbal remedies marketed without
proper quality control measures in place[5].
Herbalife is not specifically implicated in the referenced article, yet
the authors imply that Herbalife product contamination and lack of
quality control contributed to the liver injury. The authors’ assumption
is wrong and does not take into consideration that the United States
FDA requires dietary supplement manufacturers to use current Good
Manufacturing Practices (cGMPs) in the production of dietary
supplements[4].
The goal of these regulations is to “ensure that a dietary supplement
contains what the manufacturer intends” and meets specifications to
ensure the dietary supplement contains the correct ingredient, purity,
strength and composition intended. Herbalife has rigorous processes in
place concerning quality control, including extensive safety reviews
based on existing literature for product ingredients, testing to confirm
that labeled ingredients are present in finished goods, and to assure
all tested ingredients meet product specifications on an ongoing basis.
In addition to complying with cGMP regulations, Herbalife acts in
accordance with other generally recognized industry standards or
requirements by sourcing and testing raw materials to further ensure
that the final product complies with specifications for identity,
purity, potency and contaminants.
The authors also try to implicate the Camellia sinensis (C. sinensis) used in Herbalife’s tea drink products by citing case reports of liver injury in association with ethanolic extracts of C. sinensis, which contain a concentrated fraction of EGCG[1].
The most important safety consideration for green tea is the extraction
method. The historical data supporting the safety of green tea is based
on the consumption of an aqueous extract over thousands of years,
specifically, the typical three cups per day that are commonly consumed
in Asian countries. Aqueous extracts of green tea are quite different
from solvent extractions, which are commonly used to concentrate select
fractions of green tea, such as EGCG or caffeine. Again, the WJH
authors have not considered the clinical significance of potential
differences in raw material processing amongst manufacturers, controls
for contamination and identification of raw materials, and the
implication of these differences when reviewing published case reports
of liver injury. In addition, the authors state that Herbalife has
refused to provide detailed analyses of ingredients and formulations,
although no attempt was made by these authors to contact Herbalife to
obtain further information regarding Herbalife products or ingredients.
Herbalife has, to date, remained compliant with all formal regulatory
requests and requirements for product information.
The authors state that significant liver injury induced by herbal supplements is a rare event[1].
This statement is true as approximately 20 to 50 percent of all cases
presenting as hepatotoxicity are cryptogenic leading to the incidental
association of liver disease with a group of products in the absence of
specific evidence[5].
While this disease is the most common cause of drug withdrawal during
post-marketing surveillance, it is an uncommon cause of liver disease.
The background incidence of hepatotoxicity in populations is clearly
comparable to the reported incidence of immunoallergic and
individualistic reactions to allergens in foods, supplements, or the
environment. For example, in a study of 71 000 North Americans in 1992,
the background rate of idiopathic or cryptogenic liver disease was 24
cases per 100 000 individuals compared to 14 per 100 000 attributed to
cases of hepatitis B, 25 per 100 000 due to alcoholism, and 7 per
100 000 to other viral illnesses[6].
While the spectrum of liver diseases may well have changed since 1992
when this survey was done, idiopathic liver disease remains a
significant percentage of all cases. Therefore, it is particularly
important in making such associations to have incontrovertible evidence
such as is often available for prescription drugs where, under
controlled conditions, a cause-effect relationship can be established.
Finally,
the authors also state that existing case reports of dietary
supplement-induced hepatotoxicity include patients with pre-existing
liver disease and that weight loss supplements could worsen such
conditions in these patients. However, this effect could occur from many
different substances, including over-the-counter and prescription
medications, as these patients may be “pre-sensitized” due to an
underlying hepatic condition.
In conclusion, the
reference to Herbalife products as contaminated and generally comparable
to all dietary supplements or weight loss products is not
scientifically supported. Further information regarding patient
histories, concomitant medications and other compounds, dechallenge
results, and product specifications and usage is indicated to assess
fully the association of Herbalife products in the WJH case
reports. Therefore, the article does not objectively support a causal
relationship between the reported cases of liver injury and Herbalife
products or ingredients.
Footnotes
Peer
reviewers: Jordi Muntan¨¦, PhD, Unidad de Investigacion, Hospital
Universitario Reina Sof¨ªa, Av. Men¨¦ndez Pidal s/n, Cordoba 14004,
Spain
S- Editor Zhang SJ L- Editor Hughes D E- Editor Zheng XM
References
1. Chen
GC, Ramanathan VS, Law D, Funchain P, Chen GC, French S, Shlopov B,
Eysselein V, Chung D, Reicher S, et al. Acute liver injury induced by
weight-loss herbal supplements. World J Hepatol. 2010;2:410–415. [PMC free article] [PubMed]
2. Stickel
F, Droz S, Patsenker E, Bögli-Stuber K, Aebi B, Leib SL. Severe
hepatotoxicity following ingestion of Herbalife nutritional supplements
contaminated with Bacillus subtilis. J Hepatol. 2009;50:111–117. [PubMed]
3. Biotechnology Program Under the Toxic Substances Control Act (TSCA) Bacillus subtilis Final Risk Assessment; 1997. Feb, Available from:
http: //www.epa.gov/biotech_rule/pubs/fra/fra009.htm.
4. De Smet PA. Herbal remedies. N Engl J Med. 2002;347:2046–2056. [PubMed]
5. Implementation of FDA's Current Good Manufacturing Practices for Dietary Supplements. [cited 2007 Oct 24]. Available from:
http://www.fda.gov/Food/DietarySupplements/GuidanceComplianceRegulatoryInformation/RegulationsLaws/ucm174152.htm#slide.
6. Walker AM, Cavanaugh RJ. The occurrence of new hepatic disorders in a defined population. Post-marketing Surveill. 1992;6:107–117.
Do Herbalife Products Cause Hepatitis?
Multiple medical resources have found that there are 2,000,000 people around the world who are offering products that may make you very sick.

Herbalife
is a publicly traded company based out of Los Angeles, California.
Since 1980 it has allowed people the opportunity to become independent
sales reps with the opportunity to lead others to do the same in the
form of multi-level marketing, or network marketing.
Like any large organization with several decades of experience, there are some controversies behind this one. For example, in 1986, the state of California claimed that the company made inflated claims regarding income potential for which Herbalife settled for $850,000, and in 2004 thousands of current and former distributors took the company to court on the grounds of running a pyramid scheme. Herbalife settled the latter case for $6,000,000 (less than $700 each).
Along with these legal matters are much more pressing health concerns. Several medical resources have found that Herbalife products may actually cause hepatitis.
What is Hepatitis and How is it Caused?
Hepatitis can be characterized as an inflammation of the liver. One visible symptom is jaundice, and less visible ones are lack of appetite and a feeling of overall discomfort.
Hepatitis is typically caused by a virus, but it can also be caused by toxins when taken regularly, such as alcohol or an infection that is already present. The question on many people's minds regarding Herbalife products would be, how could these products be linked to infecting those who take them?
The answer is in the ingredients. There has to be something toxic in some or all of the products released by the company, but it is hard to know which ones because the FDA does not regulate herbal supplements the same way it does medicine. This is why athletes find out via heart attack that ephedrine is not so great, and how health conscious people discover that they may be poisoning themselves the hard way, as reported in a 2007 article published in The Journal of Hepatology titled "Herbal Does not Mean Innocuous."
Countries That Have Reported Illness Links to Herbalife
It is important to note where the sources are that have reported on Herbalife. This is because some people go on the attack against networking companies because they think they are dangerous or terrible, and some governments are anti-capitalistic, so they would be against a company like Herbalife because they do not want their people to have too much freedom and control.
In 2004, 12 patients in Israeli hospitals with severe liver problems had one thing in common. They were taking an Herbalife supplement that aided digestion. When their liver enzymes normalized, they resumed their normal regimen of the product and were sick again.
The 2007 article noted from the European Journal of Hepatology had several sources, one of which was medical professionals from Switzerland. In it, the ten most severe cases covered showed two patients with "certain causality" while the other eight had "probable causality." The duration of the illness ranged from two months to twelve years.
The objective here is not to demonize a company or industry, but to alert consumers that when Herbalife distributors pitch the quality of their products, they may be pitching high-end hepatitis. And while the ingredients linked to causing disease in the past may have been rid of, it does not mean that today's products are safe.
Without FDA interaction, humans are the test subjects for herbal supplements. The best way to go when it comes to supplements is to use one with a long track record of not hurting its users.
Jan. 5 update from the author: On December 23, George Fischer and one of his doctors had a conference call with me. During the course of that conversation, they promised that they would send me information contradicting the reporting in the Journal of Hepatology. No such contact has been made since.
Like any large organization with several decades of experience, there are some controversies behind this one. For example, in 1986, the state of California claimed that the company made inflated claims regarding income potential for which Herbalife settled for $850,000, and in 2004 thousands of current and former distributors took the company to court on the grounds of running a pyramid scheme. Herbalife settled the latter case for $6,000,000 (less than $700 each).
Along with these legal matters are much more pressing health concerns. Several medical resources have found that Herbalife products may actually cause hepatitis.
What is Hepatitis and How is it Caused?
Hepatitis can be characterized as an inflammation of the liver. One visible symptom is jaundice, and less visible ones are lack of appetite and a feeling of overall discomfort.
Hepatitis is typically caused by a virus, but it can also be caused by toxins when taken regularly, such as alcohol or an infection that is already present. The question on many people's minds regarding Herbalife products would be, how could these products be linked to infecting those who take them?
The answer is in the ingredients. There has to be something toxic in some or all of the products released by the company, but it is hard to know which ones because the FDA does not regulate herbal supplements the same way it does medicine. This is why athletes find out via heart attack that ephedrine is not so great, and how health conscious people discover that they may be poisoning themselves the hard way, as reported in a 2007 article published in The Journal of Hepatology titled "Herbal Does not Mean Innocuous."
Countries That Have Reported Illness Links to Herbalife
It is important to note where the sources are that have reported on Herbalife. This is because some people go on the attack against networking companies because they think they are dangerous or terrible, and some governments are anti-capitalistic, so they would be against a company like Herbalife because they do not want their people to have too much freedom and control.
In 2004, 12 patients in Israeli hospitals with severe liver problems had one thing in common. They were taking an Herbalife supplement that aided digestion. When their liver enzymes normalized, they resumed their normal regimen of the product and were sick again.
The 2007 article noted from the European Journal of Hepatology had several sources, one of which was medical professionals from Switzerland. In it, the ten most severe cases covered showed two patients with "certain causality" while the other eight had "probable causality." The duration of the illness ranged from two months to twelve years.
The objective here is not to demonize a company or industry, but to alert consumers that when Herbalife distributors pitch the quality of their products, they may be pitching high-end hepatitis. And while the ingredients linked to causing disease in the past may have been rid of, it does not mean that today's products are safe.
Without FDA interaction, humans are the test subjects for herbal supplements. The best way to go when it comes to supplements is to use one with a long track record of not hurting its users.
Jan. 5 update from the author: On December 23, George Fischer and one of his doctors had a conference call with me. During the course of that conversation, they promised that they would send me information contradicting the reporting in the Journal of Hepatology. No such contact has been made since.
LIVER DAMAGE IN EUROPE ASSOCIATED WITH HERBALIFE USE:
4 Sept 2007 ReviewThe following reports below of HerbaLife-associated liver failure appear on Medline – from Spain, then Israel and Switzerland.
:The July 2007 reports of the two dozen Herbalife-associated hepatitis cases from Israel & Switzerland reveal that liver problems occurred after about 5 months on the products; and that relapse occurred in about 20% on rechallenge with Herbalife ie in this percentage the association is proven.
Herbalife results for weight control have been reported as good. The only problem is historical according to the current Wikipedia entry: “Some of the original Herbalife weight loss products contained the active ingredient Ma Huang or Sida cordifolia, two herbs containing ephedrine alkaloids.
Adverse reactions involving the company’s Thermojetics original green tablets were recorded by the U.S. Food and Drug Administration and Herbalife subsequently stopped using ephedrine in its products in the face of rising insurance premiums.[3][4] The U.S. FDA banned supplements containing ephedra in 2004.[5]“
It is possible that the case reports below are unrelated to Herbalife itself , or that in those countries ephedra-containing Herbalife was still in use at the time, or that potentially hazardous herbs etc were added locally.
From Yahoo.com, there is an authoritative rebuttal from Iceland dated February 2007.
A score of drugs and herbs can cause liver damage, topical ones – albeit rarely- include mushrooms; black cohosh and kava – see a recent list.
Drugs like ticrynafen, methyldopa and cerivastatin were discarded among other reasons because of liver problems, which are among many reasons why necessary sex hormone contraception and replacement should rather not use designer patent ie synthetic drugs, and especially not by mouth (hepatic first pass effect).
So it is always difficult to blame a single product, as the ongoing debate about black cohosh shows – which many “first world” regulators have “black boxed” ie added a compulsory warning to black cohosh warnings.
As with black cohosh, with a rare adverse event report, users of such products must weigh up for themselves.
As they say, since Herbalife tends to be custom-made in each country, with numerous ingredients (some undisclosed), it is so far impossible to incriminate whether the cause was local product corruption, or some appreoved component, of which the known possible culprits are ephedra and camelia.
Other known hepatotoxic herbs like black cohosh, kava and mushrooms were not mentioned. A few of the patients had viral hepatitis. Only 7 cases had also taken other known potential liver sensitizers – some synthetic sex hormones (4), aspirin (3), statin (1) and hydrochlorothiazide(1), of which 2 cases had positive recurrence of hepatitis on rechallenge with Herbalife.
UCT Medicines Information Centre is unaware of any such problems locally, and can recollect only perhaps 2 queries about Herbalife in some 23 years. Clarification is awaited from Herbalife headquarters.
From Swiss data the estimated incidence was below 2 cases per million Herbalife users, but both studies were based only on hospital records.
Considering the severe global problem of hepatitis from other causes (due to alcohol; obesity/diabetes (steatohepatitis, sulphonylureas, glitazones); numerous infections; carbon tetrachloride; synthetic sex hormones (oral contraception and postmenopausal hormone therapy) , mushrooms, antibiotics and antivirals, , autoimmune disease, antiepileptics, nifedipine, amitryptiline, allopurinol, nonsteroidal anti-inflammatories including aspirin and paracetamol , black cohosh, kava, antifungals and paracetamol), and that the rare adverse association of herbalife with liver damage may well have been limited only to Herbalife products made in those three “European” countries at that time, there is clearly no cause for alarm about Herbalife – just awareness.
The urgent problem of endemic liver disease is rather the avoidance of infections and potentially hazardous antimicrobials; mushrooms; carbon tetrachloride, alcohol excess; sale pf paracetamol without inclusion of protective vitamins and N-acetyl cysteine; and avoidance of potential hepatotoxins which are rarely if ever justified considering their risks, and safe effective alternatives available for eg statins, sulphonylureeas, glitazones, black cohosh, kava, non-steroidal anti-inflammatories; oral sex hormones; and sulphonylureas.
The centuries-proven plant galega officinalis (extract) metformin after 85years of modern use remains the only drug proven in longterm use to both reduce liver damage, lipidemia, thrombosis, adiposity and insulin resistance, and thus almost halve the incidence of new diabetes, hypertension/vascular disease, cancer and thus all-cause premature medical mortality.
Thus appropriate general use of metformin with long-proven vitamins, minerals, biologicals, safe herbs, fish oil and systemic human sex hormones – combined with prudent lifestyle and largely natural fresh foodstuffs- – does away with most of the well-known potential hepatitis drug risks listed above.
In defence of free market enterprise and choices, those who choose convenience safe proven food substitutes or other complementary products as part of an acceptable balanced regime advocated by suppliers like Herbalife do well, they should just be sure of the ingredients and supplier; and they should report and discuss what they use with some knowledgable up-to-date healthcare provider.
Response from Herbalife
04.09.07
Herbalife’s South Africa CEO responds reassuringly:
Good day,
Herewith a statement from Herbalife in response to the issues raised by yourself earlier in the week:
While we are aware of reports of abnormal liver function blood tests such as those reported by Dr. Oneta, our extensive consultation with internationally recognised liver experts has led repeatedly to the conclusion that these associations in time cannot be linked to any Herbalife product.
These small numbers of reports are anecdotal and millions of satisfied customers all over the world have been using our products for more than 27 years. All Herbalife products are formulated and manufactured in accordance with strict standards overseen by the Herbalife Scientific Advisory Board, which is chaired by David Heber, M.D., Ph.D., F.A.C.P., F.A.C.N. Quality control is overseen by our Scientific Affairs Group, chaired by Y. Steve Henig PhD and made up of an international panel of experts in nutrition and botanical dietary supplements.
Herbalife products, which are now sold in 65 countries, are formulated, registered and labelled in accordance with the regulatory requirements in every market where sold. All Herbalife products are safe to consume as directed.
Many consumers who choose to use Herbalife weight-management products for weight loss are overweight, some significantly so. Pre-existing medical conditions such as obesity and diabetes can be associated with non-alcoholic fatty liver disease, a disorder that may return certain types of abnormal blood test results. These test results, therefore, may have nothing to do with any herbal supplement, but rather are the result of a pre-existing medical condition. In addition, it is possible for an individual to have an allergic reaction to our products, the same way one might to any food product; for example, strawberries or shellfish. Herbalife supports the recommendation that consumers visiting their doctors for medical treatment inform them of any supplements they may be taking.
As a socially responsible company, we operate an adverse event reporting procedure that deals with the small number of queries we have from doctors and consumers and we operate an open dialogue policy with the medical community. All adverse event reports are investigated thoroughly in consultation with the consumer and the physician (if they are available) to fully understand the facts. None have resulted in the compulsory withdrawal of any product, ever. In the United States, Herbalife actively lobbied Congress to pass legislation mandating the submission of all dietary supplement and over-the-counter drug serious adverse events to the Food & Drug Administration. That new law takes effect December 22, 2007.”
REFS:
J Hepatol. 2007 Oct;47(4):521-526. Herbal does not mean innocuous: Ten cases of severe hepatotoxicity associated with dietary supplements from Herbalife((R)) products. Schoepfer AM, ea.University Hospital Bern, Switzerland.
METHODS: To determine the prevalence and outcome of hepatotoxicity due to Herbalife((R)) products. A questionnaire was sent to all public Swiss hospitals. Reported cases were subjected to causality assessment using the CIOMS criteria. RESULTS: Twelve cases of toxic hepatitis implicating Herbalife((R)) preparations (1998-2004) were retrieved, 10 sufficiently documented to permit causality analysis. Median age of patients was 51 years (range 30-69) and latency to onset was 5 months (0.5-144). Liver biopsy (7/10) showed hepatic necrosis, marked lymphocytic/eosinophilic infiltration and cholestasis in five patients. One patient with fulminant liver failure was successfully transplanted; the explant showed giant cell hepatitis. Causality assessment of adverse drug reaction was classified as certain in two, probable in seven and possible in one case(s), respectively. CONCLUSIONS: We present a case series of toxic hepatitis implicating Herbalife((R)) products. Liver toxicity may be severe. A more detailed declaration of components and pro-active role of regulatory agencies would be desirable.
J Hepatol. 2007 Oct;47(4):514-520. Association between consumption of Herbalife((R)) nutritional supplements and acute hepatotoxicity. Elinav E, ea -Hebrew University Medical Center, Israel.
: In 2004, identification of four index cases of acute hepatitis associated with Herbalife((R)) intake led to a ministry of health investigation in all Israeli hospitals. Twelve patients with acute idiopathic liver injury in association with consumption of Herbalife((R)) products were investigated.
RESULTS: Eleven of the patients were females, aged 49.5+/-13.4 y. One patient had stage I primary biliary cirrhosis and another had hepatitis B. Acute liver injury was diagnosed after 11.9+/-11.1 months of initiation of Herbalife((R)) consumption. Liver biopsies demonstrated active hepatitis, portal inflammation rich with eosinophils, ductular reaction and parenchymal inflammation with peri-central accentuation.
. CONCLUSIONS: An association between intake of Herbalife((R)) products and acute hepatitis was identified in Israel. We call for prospective evaluation of Herbalife((R)) products for possible hepatotoxicity.
Med Clin (Barc). 2007 Feb 17;128(6):238-9.
[Hepatotoxicity associated with the consumption of herbal slimming products] Duque JM,ea. [Article in Spanish] Letter
==============================
J Hepatol (2007) 0: .
Herbal does not mean innocuous: Ten cases of severe hepatotoxicity associated with dietary
AM Schoepfer, A Engel, K Fattinger, UA Marbet, D Criblez, J Reichen,
A Zimmermann, CM Oneta
BACKGROUND/AIMS: Herbal agents are popular and perceived as safe because they are supposedly 'natural'. We report 10 cases of toxic hepatitis implicating Herbalife((R)) products. METHODS: To determine the prevalence and outcome of hepatotoxicity due to Herbalife((R)) products. A questionnaire was sent to all public Swiss hospitals. Reported cases were subjected to causality assessment using the CIOMS criteria. RESULTS: Twelve cases of toxic hepatitis implicating Herbalife((R)) preparations (1998-2004) were retrieved, 10 sufficiently documented to permit causality analysis. Median age of patients was 51 years (range 30-69) and latency to onset was 5 months (0.5-144). Liver biopsy (7/10) showed hepatic necrosis, marked lymphocytic/eosinophilic infiltration and cholestasis in five patients. One patient with fulminant liver failure was successfully transplanted; the explant showed giant cell hepatitis. Sinusoidal obstruction syndrome was observed in one case. Three patients without liver biopsy presented with hepatocellular (2) or mixed (1) liver injury. Causality assessment of adverse drug reaction was classified as certain in two, probable in seven and possible in one case(s), respectively. CONCLUSIONS: We present a case series of toxic hepatitis implicating Herbalife((R)) products. Liver toxicity may be severe. A more detailed declaration of components and pro-active role of regulatory agencies would be desirable.
=====================
Results of a search for studies that show the effects of using Herbalife products for weight loss.
The official website is: http://www.ncbi.nlm.nih.gov/pubmed?term=herbalife
BACKGROUND/AIMS: Herbal agents are popular and perceived as safe because they are supposedly 'natural'. We report 10 cases of toxic hepatitis implicating Herbalife((R)) products. METHODS: To determine the prevalence and outcome of hepatotoxicity due to Herbalife((R)) products. A questionnaire was sent to all public Swiss hospitals. Reported cases were subjected to causality assessment using the CIOMS criteria. RESULTS: Twelve cases of toxic hepatitis implicating Herbalife((R)) preparations (1998-2004) were retrieved, 10 sufficiently documented to permit causality analysis. Median age of patients was 51 years (range 30-69) and latency to onset was 5 months (0.5-144). Liver biopsy (7/10) showed hepatic necrosis, marked lymphocytic/eosinophilic infiltration and cholestasis in five patients. One patient with fulminant liver failure was successfully transplanted; the explant showed giant cell hepatitis. Sinusoidal obstruction syndrome was observed in one case. Three patients without liver biopsy presented with hepatocellular (2) or mixed (1) liver injury. Causality assessment of adverse drug reaction was classified as certain in two, probable in seven and possible in one case(s), respectively. CONCLUSIONS: We present a case series of toxic hepatitis implicating Herbalife((R)) products. Liver toxicity may be severe. A more detailed declaration of components and pro-active role of regulatory agencies would be desirable.
=====================
Results of a search for studies that show the effects of using Herbalife products for weight loss.
The official website is: http://www.ncbi.nlm.nih.gov/pubmed?term=herbalife















No comments:
Post a Comment